Contact Us
CONTACT
TaiwanClinicalTrials.tw is an online information platform that is established and managed by the Center for Drug Evaluation (CDE) Taiwan to promote and to provide the essential information and excellence of Taiwan’s clinical trial environment to the public, academia and industry.
- If you have any inquiry regarding clinical trial related events, information, and patient recruitments advertisement please fill out our inquiry form bellow.
- If you need consultation services regarding regulations and clinical trial design please visit the consultation service page provided by CDE Taiwan.
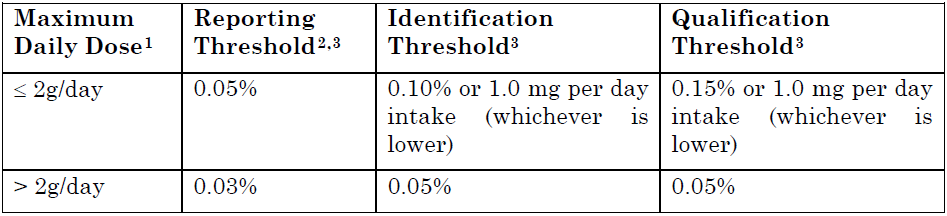
 2.Types of impurities, structural characterisation data and batch analysis data (including clinical, non-clinical, stability data, etc.) should be provided in 3.2.S.3.2. with explanations on whether these impurities are listed in the specifications.
2.Types of impurities, structural characterisation data and batch analysis data (including clinical, non-clinical, stability data, etc.) should be provided in 3.2.S.3.2. with explanations on whether these impurities are listed in the specifications.
